|
4/2/2024 0 Comments Molar masses on periodic table
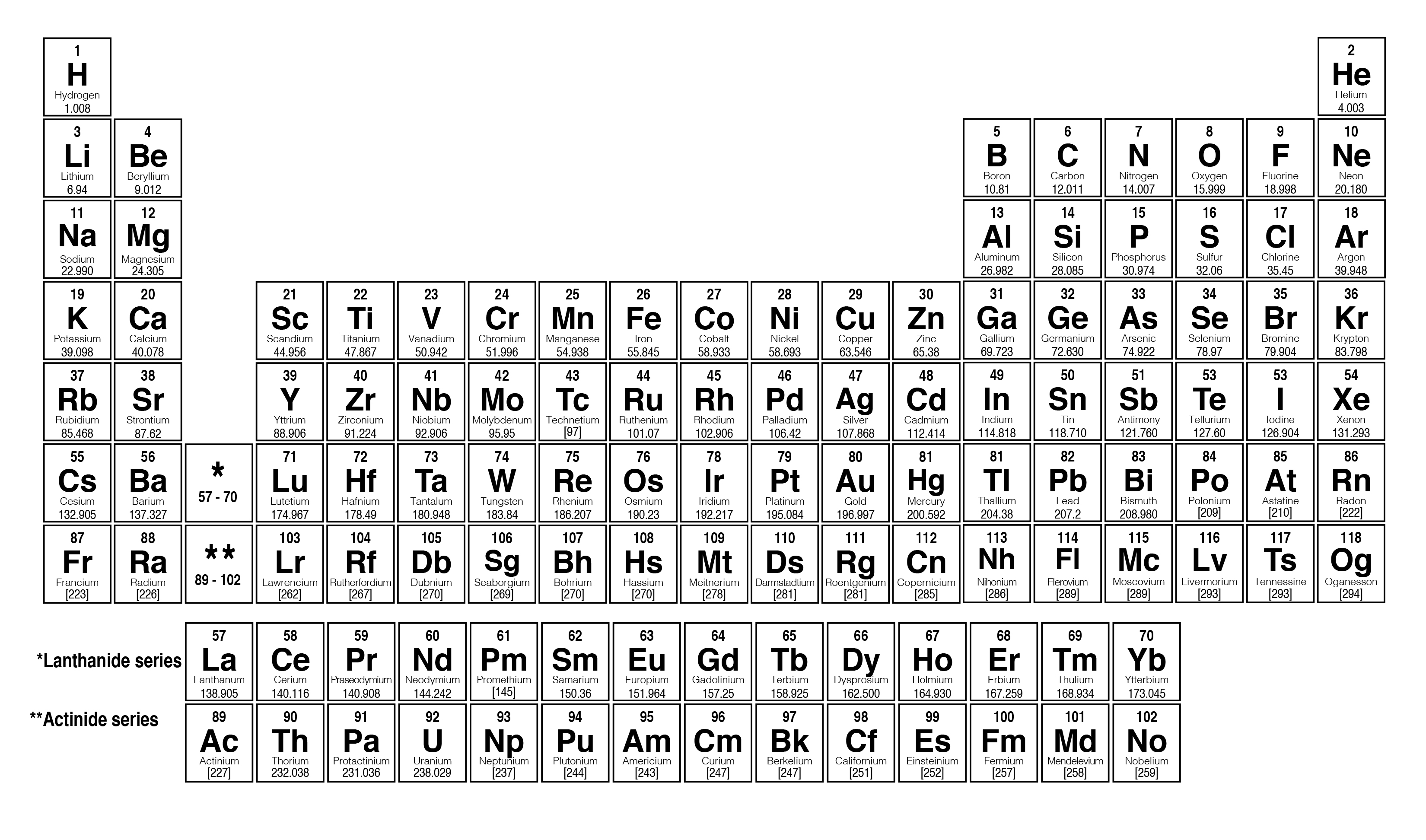
He noticed that elemental properties repeated every seventh (or multiple of seven) element, as musical notes repeat every eighth note. John Alexander Reina Newlands was an English chemist who worked on the development of the periodic table. Despite the fact that Newlands’s table had no logical place for the d-block elements, he was honored for his idea by the Royal Society of London in 1887. Molar mass can be calculated by using the periodic table and following three simple steps. At one scientific meeting, Newlands was asked why he didn’t arrange the elements in alphabetical order instead of by atomic mass, since that would make just as much sense! Actually, Newlands was on the right track-with only a few exceptions, atomic mass does increase with atomic number, and similar properties occur every time a set of ns 2 np 6 subshells is filled. 2: Variation of Atomic Volume with Atomic Number. In Meyer’s plot of atomic volume versus atomic mass, the nonmetals occur on the rising portion of the graph, and metals occur at the peaks, in the valleys, and on the downslopes. Unfortunately, Newlands’s “law of octaves” did not seem to work for elements heavier than calcium, and his idea was publicly ridiculed. 2, the alkali metals have the highest molar volumes of the solid elements. There were seven elements because the noble gases were not known at the time. 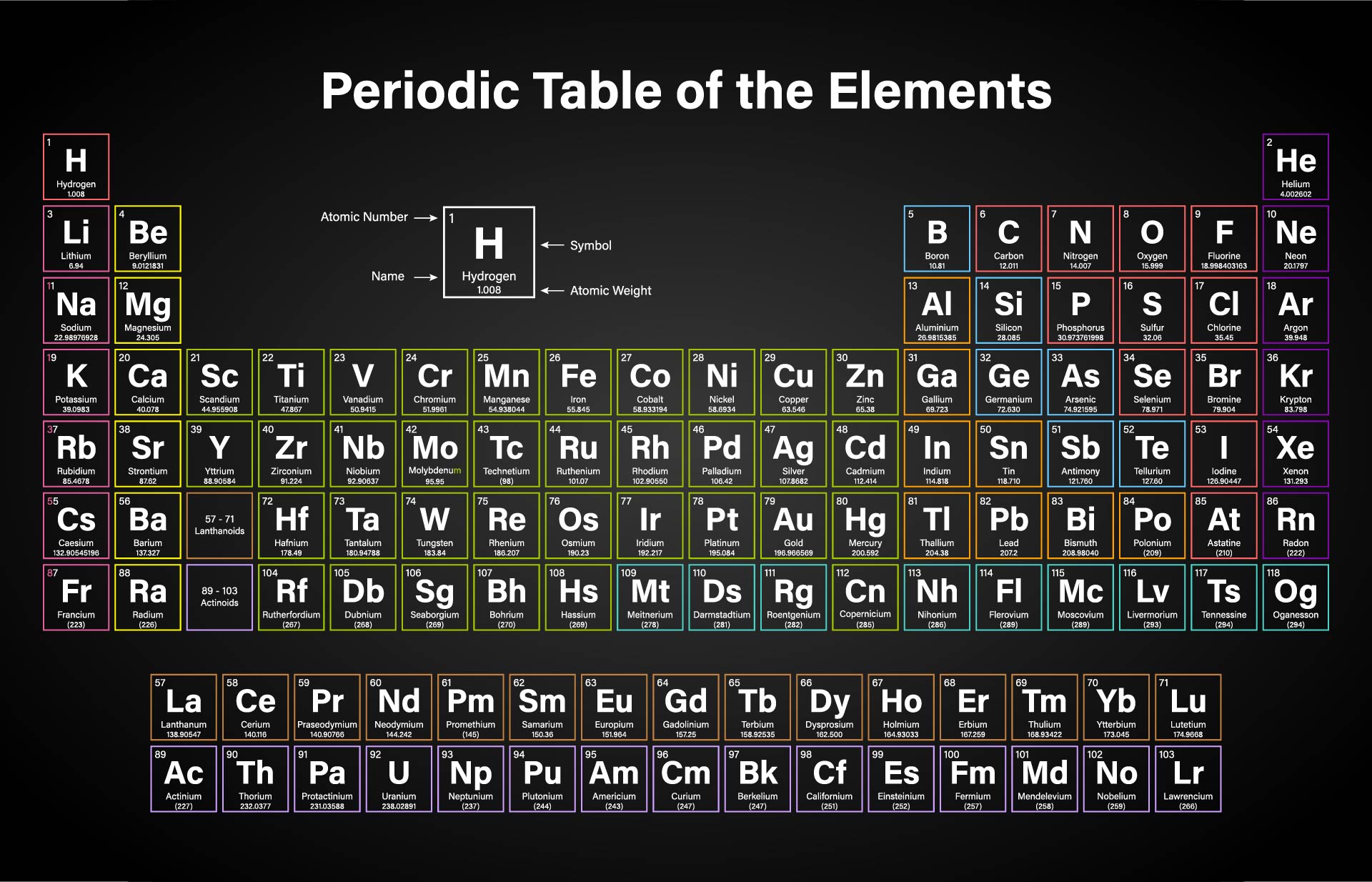
He described octaves as a group of seven elements which correspond to the horizontal rows in the main groups of today's periodic table. Newlands therefore suggested that the elements could be classified into octaves. So it is stored by being coated with petroleum jelly. It cannot be kept under oil, as sodium can, because it is less dense and floats. The lessons learned in this post will continue through the study of chemical reactions and equations.\)). Lithium is a soft, silvery-white, metal that heads group 1, the alkali metals group, of the periodic table of the elements. In chemistry, the molar mass of a compound (represented by M m / n) is the mass of the substance (in grams), divided by the number of moles of the substance. But first of all, let’s start with a simple introduction. We also learned how to calculate the molar mass of a compound using the periodic table and how to convert mass into moles. If you want to learn, how to calculate the molar mass in periodic table, you will love this step-by-step explanation. Plug these values into Equation 1 and youll find the molar mass of water to be around 18g/mole. 1.007 g/mole for Hydrogen and 15.999 g/mole for Oxygen. We learned about Avogadro’s number and how it relates to the mole, which is a unit used to express the amount of a substance. Molar mass of H 2O (2 x atomic mass of Hydrogen) + (atomic mass of Oxygen) We find the atomic mass of Hydrogen and Oxygen in the periodic table. Meyer arranged the elements by their atomic volume, which today is equivalent to the molar volume, defined as molar mass divided by molar density. In conclusion, understanding molar mass is an essential concept in chemistry as it allows us to relate the amount of a substance to the number of particles present in it. The periodic table achieved its current form through the work of Dimitri Mendeleev and Julius Lothar Meyer, who both focused on the relationship between atomic mass and chemical properties. Specifically, the number is defined as 6.022 \times 10^.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
